Summary
- Effective pharmaceutical waste depackaging solutions can decrease hazardous waste by up to 70% while ensuring adherence to EPA, DEA, and state regulations.
- Different pharmaceutical waste streams necessitate specific handling procedures—hazardous medications must be kept separate from non-hazardous ones to avoid contaminating the environment.
- Automated depackaging technology can greatly reduce labor costs and compliance risks compared to manual processes in healthcare facilities.
- Healthcare facilities could face fines of up to $70,000 per day for improper pharmaceutical waste management under RCRA regulations.
- Stericycle offers comprehensive depackaging solutions that assist healthcare facilities in maintaining compliance while minimizing their environmental impact.
The Pharmaceutical Waste Crisis: The Importance of Proper Depackaging

“Reducing Medical Waste Repurpose Health …” from uplink.weforum.org and used with no modifications.
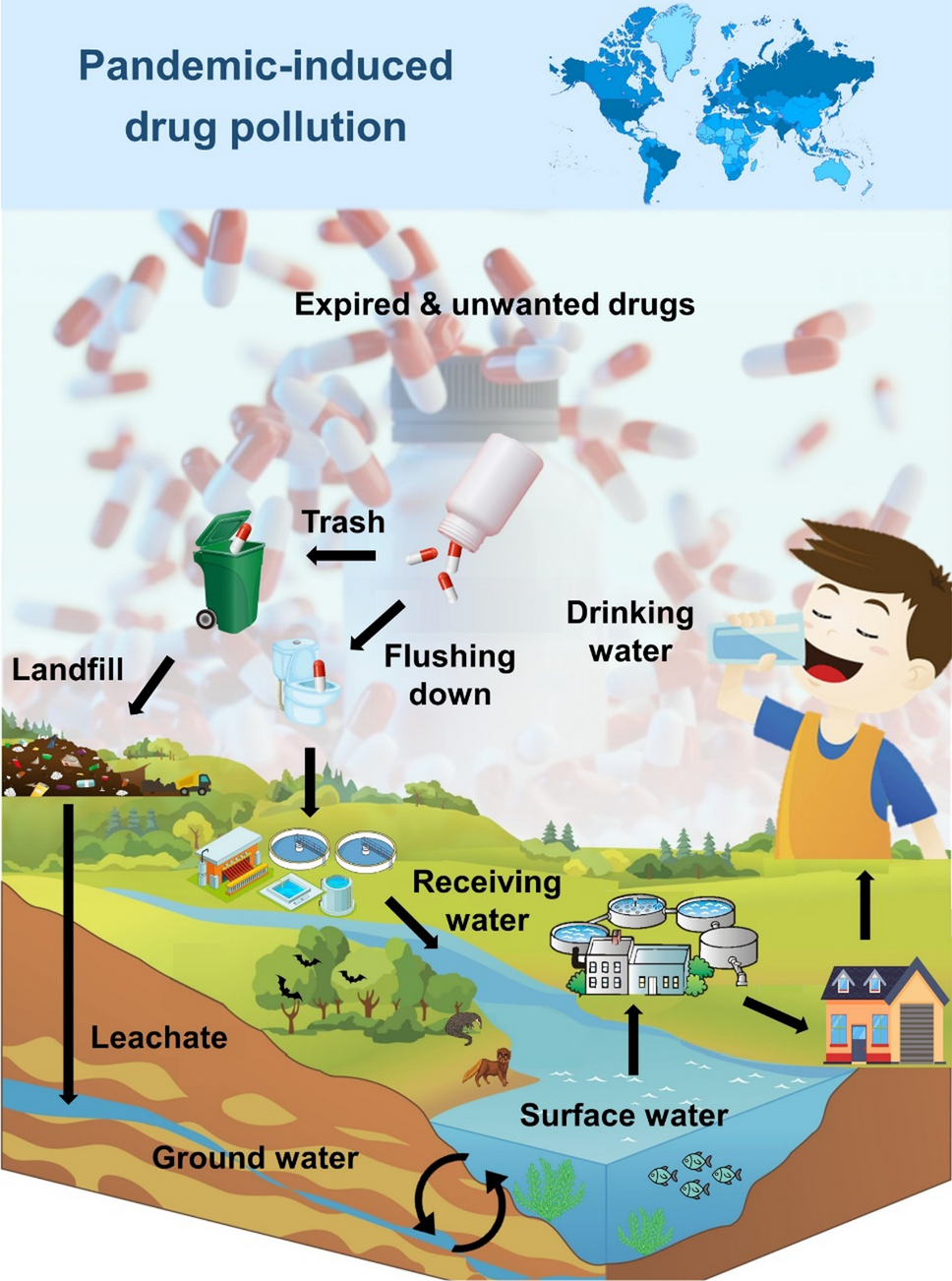
Healthcare facilities across North America produce over 5 billion pounds of waste each year, with pharmaceutical waste being one of the most difficult components to manage. Proper depackaging—the act of removing medications from their packaging before disposal—is crucial for regulatory compliance and environmental protection. When pharmaceuticals are left in their packaging, they often cannot be properly treated, leading to water system contamination and potential public health risks.
Depackaging is not as simple as taking pills out of a bottle. It’s a systematic process that makes sure dangerous elements are found, separated, and thrown away according to strict rules and regulations. Without the right depackaging solutions, healthcare facilities could face big fines, harm the environment, and damage their reputation. Stericycle has created special processes that help facilities deal with these complicated rules while also reducing the amount of waste they produce.
Recent studies have revealed that pharmaceutical compounds are present in drinking water sources nationwide, which is a significant concern. These contaminants are mainly the result of medications that have been disposed of improperly, without being correctly depackaged and treated. According to the EPA, if medications are not properly depackaged before disposal, up to 80% of the medication compounds can end up in our waterways.
Special Handling for Different Types of Pharmaceutical Waste
“Waves of pharmaceutical waste …” from link.springer.com and used with no modifications.
“Pharmaceutical waste is not uniform—it ranges from relatively benign expired medications to highly toxic chemotherapy agents. Each category requires different handling procedures, and mixing these streams creates compliance risks and increases disposal costs.” — Environmental Protection Agency, 2022 Waste Management Guidelines
It’s essential to understand the diverse nature of pharmaceutical waste to put effective depackaging solutions in place. Each waste stream comes with its unique challenges and requires specific protocols to ensure regulatory compliance and environmental protection. The complexity increases with the variety of medications used in modern healthcare settings, from tablets and capsules to patches, creams, and liquid formulations.
Another layer of complexity comes from the packaging itself. Blister packs, bottles, vials, and unit-dose packaging each require unique separation approaches. Many facilities overlook the technical challenges involved, especially when handling controlled substances that must retain chain-of-custody documentation throughout the depackaging process.
Especially for bigger facilities, the amount of waste can be mind-boggling. For example, a 250-bed hospital typically produces about 7,000 pounds of pharmaceutical waste each year. Without effective depackaging solutions, handling this amount of waste becomes laborious and prone to mistakes, thereby raising the likelihood of non-compliance and environmental damage.
Understanding the Difference Between Hazardous and Non-Hazardous Pharmaceutical Waste

“Hazardous Vs. Non-Hazardous Waste – Key …” from cerclex.com and used with no modifications.
Knowing the difference between hazardous and non-hazardous pharmaceutical waste is crucial for setting up a compliant disposal program. Pharmaceuticals can be classified as hazardous waste under RCRA regulations if they are toxic, ignitable, corrosive, or reactive. Some examples are warfarin, nicotine products, chemotherapy agents, and many hormonal medications. Special handling is required during depackaging to prevent contamination of the environment and worker exposure. For more information on how to manage hazardous pharmaceutical waste, you can read about reducing hazardous waste with compliance packaging.
Non-hazardous pharmaceutical waste, while less regulated, still requires proper management. This category includes most antibiotics, non-toxic over-the-counter medications, and many prescription drugs. Though not classified as hazardous under federal regulations, many states and localities have implemented stricter requirements for these substances based on growing evidence of their environmental impact. Effective depackaging solutions must account for both waste streams while maintaining clear separation throughout the process.
Managing Controlled Substances and Meeting DEA Requirements
“What is a Controlled Substance …” from cpdonline.co.uk and used with no modifications.
Controlled substances pose specific problems in the depackaging process because they are closely regulated by the Drug Enforcement Administration. These drugs, which include opioids, stimulants, and some types of sedatives, must be made non-retrievable using specific destruction methods. The depackaging of controlled substances requires witnesses, secure processing areas, and detailed record-keeping that tracks each unit from the pharmacy to its final destruction. The penalties for non-compliance are severe, including hefty fines and the possibility of criminal charges for those responsible.
Non-prescription Drugs and Supplements
Non-prescription drugs and dietary supplements make up a large amount of pharmaceutical waste in many facilities. While these are often seen as less dangerous, some non-prescription drugs contain ingredients that make them regulated waste. Products that have a high concentration of iron, liquids based on alcohol, and certain topical preparations may need special handling when being unpackaged. Non-prescription drugs come in a wide variety of packaging formats, from blister packs to bottles and tubes, so depackaging solutions need to be able to handle all of these efficiently and be able to correctly classify the waste streams that result.
Managing over-the-counter (OTC) waste is a significant challenge, especially considering the vast amounts of these products in circulation. Healthcare facilities, particularly long-term care institutions, often generate substantial amounts of expired or discontinued OTC medications. It’s important to have depackaging processes that can effectively handle these quantities while maintaining regulatory compliance and environmental responsibility.
Legal Guidelines for Pharmaceutical Waste Disposal
There are a variety of federal, state, and local laws that healthcare facilities must adhere to when it comes to pharmaceutical waste disposal. These laws are constantly changing as we continue to learn more about the impact of pharmaceuticals on the environment. What was acceptable five years ago might not be acceptable today, which is why it’s so important to stay educated and keep waste management programs up to date.
There are significant financial implications, with potential fines reaching up to $70,000 per day in certain situations. This doesn’t even take into account the potential damage to reputation and possible legal actions. Many facilities underestimate the extent of these regulations, focusing mainly on controlled substances while neglecting equally important RCRA hazardous waste requirements and state-specific mandates.
Understanding EPA and RCRA Requirements

“Recovery Act (RCRA) Regulations …” from www.epa.gov and used with no modifications.
The Resource Conservation and Recovery Act (RCRA), enforced by the Environmental Protection Agency (EPA), is the main federal law that deals with the management of pharmaceutical waste. This law states that pharmaceuticals that are toxic, ignitable, corrosive, or reactive, or that are on certain P and U lists, must be treated as hazardous waste. In 2019, the EPA released the Management Standards for Hazardous Waste Pharmaceuticals rule. This rule, which specifically addresses healthcare facilities, makes it illegal to dispose of hazardous waste pharmaceuticals in the sewer system. It also provides specific standards for managing these types of waste.
Meeting RCRA compliance means that facilities must correctly identify, separate, and document any hazardous pharmaceutical waste during the depackaging process. This involves keeping accurate waste determination records, training staff on the correct identification procedures, and putting systems in place that stop hazardous and non-hazardous streams from cross-contaminating. Facilities also have to make sure that their depackaging methods don’t accidentally release hazardous components into the environment. This requires controlled environments and suitable containment measures.
DEA Rules for Controlled Drugs
The Drug Enforcement Administration’s rules as part of the Controlled Substances Act add another level of compliance requirements for drugs that are classified as Schedules I to V. The 2014 Secure and Responsible Drug Disposal Act and the rules that followed set specific protocols for disposing of controlled substances. These drugs must be made “non-retrievable” through methods of destruction that cannot be reversed. Solutions for depackaging that are compliant with the DEA must maintain strict documentation of the chain-of-custody, use secure areas for processing, and often require that multiple authorized personnel witness the process.
Facilities depackaging controlled substances that may also qualify as RCRA hazardous waste face unique challenges due to the intersection of DEA and EPA regulations. These “dual-regulated” medications must meet requirements for both witnessed destruction and hazardous waste management at the same time. This requires specialized processes and documentation systems. If these overlapping requirements are not properly navigated, facilities can face penalties from multiple regulatory agencies. For more insights on managing such waste, you can explore compliance packaging strategies.
Requirements That Vary By State
Healthcare facilities often have to comply with state regulations that are more stringent than federal requirements. For instance, the Medical Waste Management Act in California sets a higher bar for handling pharmaceutical waste than federal regulations, necessitating specific containment and treatment techniques for all pharmaceutical waste, irrespective of its federal hazardous classification. Connecticut, Washington, and Minnesota have also broadened their definitions of regulated pharmaceutical waste, necessitating thorough depackaging solutions to manage these wider waste streams.
It can be difficult to standardize procedures due to the different regulatory requirements in various regions. This is particularly true for healthcare systems that operate in several states. Depackaging programs must be adaptable to these regional variations while maintaining operational efficiency. Some states also have pharmaceutical take-back programs that affect how facilities handle certain waste streams. These programs must be integrated with depackaging protocols.
Medical Center Accreditation Standards
“Hospital Accreditation Standards …” from fastercapital.com and used with no modifications.
Aside from government regulations, additional pharmaceutical waste management requirements are imposed on medical centers by accrediting bodies such as The Joint Commission, DNV GL Healthcare, and the Healthcare Facilities Accreditation Program. These organizations assess waste management practices during facility inspections, with deficiencies potentially affecting accreditation status. Accreditation standards often stress staff training, documentation consistency, and program effectiveness, along with technical compliance with regulations.
Choosing the Right Depackaging Methods for Various Types of Waste
When deciding on the best depackaging methods, it’s essential to consider the type of waste, the capabilities of the facility, and any regulatory requirements. There is no one-size-fits-all solution in pharmaceutical waste management as each type of medication comes with its own set of challenges. Today’s depackaging systems usually include a combination of manual and automated processes, which are applied to different types of waste depending on the amount of waste, the level of hazard, and the requirements for handling the waste.
How well depackaging is done can affect both how well you follow the rules and how much you spend. Studies show that places that use the best depackaging methods can cut the number of people they need by as much as 65% and follow the rules over 40% better than places that don’t have a plan. This can save a lot of money and lower the chance of something going wrong in the long run.
Hand Depackaging Methods
Hand depackaging is still a vital part of managing pharmaceutical waste, especially for waste streams with a smaller volume and specialized medication types. This method usually requires trained staff to physically remove medications from their packaging in a controlled setting. Successful hand depackaging needs standard operating procedures that are well-documented, suitable personal protective equipment, and engineered controls to reduce exposure risks. Despite advancements in technology, certain medication types—especially multi-component products and custom-compounded preparations—still need to be processed by hand because of their complexity. For more information on the benefits of depackaging, see our article on food depackaging benefits.
Automated Depackaging Solutions
Automated depackaging solutions have brought about a new era of pharmaceutical waste management, particularly for high-volume facilities. These systems range from semi-automated units that help with specific tasks to fully integrated systems that can process thousands of dosage units per hour. High-tech features include barcode scanning for automatic documentation of disposed medications, artificial intelligence for waste stream classification, and containment systems that reduce worker exposure to hazardous components. The return on investment for these systems can be significant, with payback periods typically ranging from 12-24 months for medium to large healthcare facilities, based on labor savings alone.
When choosing automated solutions, facilities should take into account whether the solution is suitable for their specific waste streams, the space they have available, the solution’s throughput capabilities, and how it will integrate with their current waste management systems. The best technologies have modular designs that can be scaled up or down as the facility’s needs change, ensuring that they won’t become obsolete if waste volumes change. The maintenance requirements and reliability of the solution should also be important factors in the decision-making process, as any downtime could have a significant effect on compliance and operations.
Managing Liquid Medication
Liquid medications come with their own unique set of challenges in the depackaging process. This is due to the potential for splashing, aerosolization, and container residues. To effectively handle liquid medications, it’s necessary to use specialized containment systems, absorbent materials, and often solidification agents to convert liquids to solid form before final disposal. The concentration and hazard profile of the medication determine specific handling requirements. Cytotoxic and hazardous liquids require enhanced engineering controls beyond those needed for non-hazardous formulations. Many facilities implement dedicated liquid processing stations. These stations have appropriate ventilation, spill control measures, and personal protective equipment to manage these waste streams effectively.
Processing of Aerosols and Inhalers
Due to the pressurized propellants contained in aerosols and inhalers, they can pose a physical hazard during the depackaging process. In addition, these products may contain active pharmaceutical ingredients that are classified as hazardous waste. Specialized depackaging technologies for these items usually include puncturing mechanisms that safely release propellants through carbon filtration systems, then separate the medication components from the delivery devices. Propellants often qualify as hazardous waste due to their ignitability characteristics, which necessitates proper documentation and downstream management. Facilities that process large volumes of these items should provide specific training on fire safety and the pressure hazards associated with incorrect handling.
Managing Patch and Transdermal Products
Transdermal patches are among the most difficult pharmaceutical waste streams to manage because they often contain active ingredients even after a patient has used them. Many patches, including those for fentanyl, hormones, and nicotine, retain a significant amount of medication that may be considered hazardous waste under RCRA regulations. To effectively depackage patches, it’s important to use specific folding techniques to contain active surfaces, select the right containers to prevent the transfer of medication, and clearly identify the patches so that downstream handlers understand the potential hazards. Some facilities use specialized destruction technologies, such as digesters specifically for medications, to neutralize active ingredients before final disposal.
Transdermal products are varied and diverse, which means that waste stream-specific protocols should be developed rather than generic approaches. Facilities should develop illustrated guides that show the correct handling techniques for commonly used patches. The focus should be on controlling contact with adhesive surfaces that contain active pharmaceutical ingredients. Staff training should address common misconceptions about “used” patches. The training should emphasize that the medication is still active even after the patient has applied it.
Collaborative Service Models
Smaller healthcare facilities can take advantage of collaborative service models to gain access to advanced depackaging solutions without shouldering the full cost of implementation. These cooperative models allow multiple facilities to make use of a central depackaging service run by either a third party or the largest member of the collaborative group. By combining waste volumes, participants can reach the necessary economies of scale for more advanced technologies, while costs are distributed proportionally based on usage. Some hospital systems have reported cost savings of 30-40% through regional consolidation of pharmaceutical waste depackaging.
Funding and Grant Opportunities
There are many grant programs that are specifically designed to help improve healthcare waste management practices. The Pollution Prevention (P2) Grant Program from the EPA provides funding for projects that aim to reduce the amount of hazardous materials in healthcare settings, including initiatives to reduce pharmaceutical waste. In addition, many state environmental agencies run their own grant programs that help healthcare facilities transition to waste practices that are more sustainable. Many of these funding opportunities specifically cover the costs of acquiring depackaging equipment and implementing the program.
Aside from government subsidies, numerous healthcare facilities have effectively utilized community foundation funding and pharmaceutical manufacturer stewardship programs to balance the costs of implementation. When applying for these opportunities, it is important to highlight both the environmental advantages and the potential community health improvements that can result from proper pharmaceutical waste management. Funding applications are significantly strengthened by quantifying waste reduction metrics and the potential prevention of waterway contamination.
Environmental Consequences and Sustainability Factors
Pharmaceutical waste’s environmental impact goes far beyond simply meeting regulations. Research has found traces of pharmaceutical substances in surface water, groundwater, and even drinking water sources throughout North America, with certain drugs being resistant to standard water treatment methods. Properly executed depackaging solutions are a crucial first move in stopping these environmental contaminations by making sure medications receive the right treatment before they are disposed of.
Advanced depackaging programs can also play a major role in sustainability initiatives by salvaging recyclable materials that would otherwise be discarded as waste. Plastics, paper, and metal components from pharmaceutical packaging can make up as much as 60% of the total waste volume. By separating these materials through efficient depackaging, facilities can keep a significant amount of their waste out of landfills and potentially make money through recycling programs. Top healthcare facilities are now including pharmaceutical waste metrics in their sustainability reports, acknowledging the major environmental impact of these programs.
Energy-from-Waste Initiatives
Today’s energy-from-waste technologies offer a greener alternative to landfill disposal for many components of pharmaceutical waste. These systems transform non-recyclable packaging materials and certain medication residues into energy via regulated incineration processes that capture heat for electricity generation. Correctly depackaged pharmaceutical waste often yields more energy than mixed medical waste due to its more uniform composition and higher combustible content. When considering energy-from-waste options, facilities should prioritize systems that use advanced emission controls that go beyond regulatory requirements, ensuring that hazardous components are not merely transferred from solid waste to air emissions.
Recycling Valuable Materials
Depackaging solutions can be used to recover valuable materials from pharmaceutical packaging. Aluminum blister packs, glass vials, plastic containers, and cardboard packaging can all be recycled if they are properly separated from the medication components. Some depackaging systems use automated sorting technologies that separate materials by type. This can increase the amount of materials that can be recycled and reduce the rate of contamination. The materials that are recovered can be sold, which can help to offset the cost of the depackaging program. This also supports the principles of the circular economy. If a facility is starting a new depackaging program, it is important to establish relationships with recyclers who specialize in handling healthcare materials. This can help to maximize the amount of materials that are recovered.
Strategies for Reducing Carbon Footprint
The carbon footprint of managing pharmaceutical waste can be traced back to its lifecycle, from production to disposal. By depackaging strategically, the carbon footprint can be reduced significantly through several ways. Firstly, depackaging allows waste to be segregated properly, which means that treatment methods can be sized correctly, preventing the over-treatment of components that are not hazardous. Secondly, the carbon footprint of producing new packaging can be reduced by recycling packaging materials. Lastly, some facilities have implemented depackaging programs that keep track of the carbon metrics associated with different disposal pathways, which allows data-driven decisions to be made, reducing the overall impact on the climate while maintaining regulatory compliance. Compared to traditional methods, these strategies can reduce the carbon footprint of managing pharmaceutical waste by 30-50% when fully implemented.
What’s Next for Managing Pharmaceutical Waste
Pharmaceutical waste management is a rapidly changing field. New technologies, regulations, and environmental concerns are all influencing its evolution. New depackaging technologies use artificial intelligence to identify medications and automatically determine how to classify the waste. This reduces human error and improves compliance rates. Blockchain documentation systems are being tested to provide unchangeable records of how pharmaceutical waste is handled. This is especially useful for controlled substances and high-risk medications. These innovations could reduce costs and improve compliance outcomes at the same time.
In the future, we can expect to see pharmaceutical waste management become an integral part of larger sustainability efforts. The top healthcare organizations are already including pharmaceutical footprint metrics in their environmental, social, and governance (ESG) reporting frameworks. With pharmaceutical pollution increasingly being recognized as an environmental justice issue that impacts vulnerable communities, facilities that take the initiative to implement comprehensive depackaging solutions will have a leg up from both regulatory and reputational standpoints. The combination of regulatory pressure, technological innovation, and sustainability requirements makes this the perfect time for healthcare facilities to reassess and improve their pharmaceutical waste depackaging strategies.
Common Questions
Healthcare professionals often have questions about pharmaceutical waste management. They want to understand the complicated regulations and find the most efficient methods. The following are the most common questions we receive from facilities that are starting or improving their depackaging programs. The answers are based on current regulatory interpretations and the best practices in the industry. However, facilities should always work with qualified environmental compliance professionals when they create their specific programs.
Many facilities find it challenging to implement depackaging programs due to staff resistance to new processes and integration with existing workflows, beyond the technical aspects of depackaging. Successful programs usually include change management strategies that highlight the environmental and safety benefits while providing extensive training and support during the transition period. Regular feedback mechanisms enable continuous improvement and help pinpoint process obstacles before they affect compliance outcomes.
Regulatory requirements lay the groundwork for pharmaceutical waste management, but many leading organizations are going above and beyond these requirements. These organizations are using innovative approaches that focus on reducing waste at the source by managing formularies, controlling inventory, and adopting prescribing practices that limit the generation of medication waste. When these practices are paired with effective depackaging solutions, they create thorough pharmaceutical stewardship programs that address the entire lifecycle of medications in healthcare settings.
“Top-performing pharmaceutical waste management programs don’t view compliance as the finish line, but rather as the starting block. True success comes from incorporating depackaging solutions into a larger vision of environmental responsibility and operational efficiency.” – Healthcare Waste Institute, 2023 Best Practices Guide
How long should pharmaceutical waste disposal records be kept?
Pharmaceutical waste disposal records should be kept for at least three years under standard RCRA requirements, although many states require longer retention periods of up to five years. For controlled substances, DEA regulations require destruction records to be kept for two years. However, industry best practice suggests keeping all pharmaceutical waste documentation for at least five years regardless of regulatory minimums, with some risk management experts suggesting retention until seven years after disposal. This longer timeframe protects facilities during delayed regulatory actions and provides documentation for potential future environmental liability claims. Electronic record systems with appropriate security controls can make it easier to comply with these longer retention requirements and can make the records more accessible during regulatory inspections.
Are the depackaging methods for controlled substances the same as non-controlled medications?
Controlled substances necessitate different depackaging protocols that adhere to DEA compliance throughout the process. The depackaging equipment may be the same, but controlled substances must be processed under direct supervision with two witnesses documenting the destruction process. The depackaging must make the medications “non-retrievable” according to DEA standards, which usually requires more thorough destruction than non-controlled medications. Furthermore, depackaging of controlled substances must take place in secure areas with limited access and constant monitoring. Many facilities schedule specific processing times for controlled substances to keep them separate from regular pharmaceutical waste, with specialized documentation tools that meet DEA record-keeping requirements while integrating with larger waste management systems.
What type of personal protective equipment should staff handling pharmaceutical waste wear?
Personal protective equipment for pharmaceutical waste depackaging should be chosen based on a detailed risk assessment of the specific waste streams being processed. Staff generally need to wear nitrile gloves (two pairs when handling hazardous pharmaceuticals), fluid-resistant gowns, eye protection, and possibly respiratory protection when handling powders or medications that pose inhalation risks. When processing hazardous pharmaceuticals as defined by USP <800>, additional controls may be necessary, such as gloves rated for chemotherapy and closed-system processing equipment. The PPE program should cover not only routine operations but also spill response scenarios, with appropriate decontamination protocols established for each waste category. Regular fit testing and PPE inspections should be documented as part of the overall compliance program, with a special focus on ensuring glove compatibility with the specific medications being handled.
How can we tell if a drug is classified as hazardous waste?
There are several steps involved in figuring out if a drug is considered hazardous waste. The first step is to see if the drug is on the EPA’s P or U lists of hazardous chemicals. These lists include drugs such as warfarin (P001) and cyclophosphamide (U058). If the drug isn’t on these lists, the next step is to see if it has any hazardous characteristics. These include ignitability (a flash point of less than 140°F), corrosivity (a pH of less than 2 or more than 12.5), reactivity, or toxicity (failing TCLP testing). Many facilities make this process easier by using third-party databases or specialized software. These tools keep track of the current hazardous waste determinations for commercial drugs.
Facilities must make their own hazardous waste determinations for formulations that are not in standard databases. They must use safety data sheet information, manufacturer guidance, and possibly laboratory testing. As regulations change, these determinations must be documented and reviewed periodically. Some states have expanded the definition of hazardous waste beyond federal standards. This is especially true for categories such as endocrine disruptors and antibiotics. Facilities must include these additional criteria in their evaluation processes.
Are there any drugs that can’t be depackaged and must be thrown away in their original containers?
Some pharmaceutical formulations should be kept in their original containers when they’re thrown away for safety and environmental reasons. These include ampules of arsenic trioxide, which are extremely toxic and dangerous if they break; pressurized containers that can’t be safely depressurized; radiopharmaceuticals that are regulated by the Nuclear Regulatory Commission; and drugs that manufacturers specifically say should be thrown away in their original containers. Also, some facilities choose to keep very potent drugs like fentanyl patches in their original containers with extra containment to prevent exposure when they’re handled. When drugs are kept in their original containers, the reason for this exception to the normal depackaging rules should be documented appropriately.
In addition to these particular instances, a risk assessment could pinpoint other products that pose intolerable risks during the depackaging process. A team of specialists from the fields of pharmacy, safety, and waste management should make this decision, and the decision-making process should be well-documented. If medications are still in their original containers, those handling the waste downstream must be informed to ensure they use the correct treatment methods. This is because some treatment technologies are designed for depackaged waste streams.
Advancements in technology and environmental consciousness are continuously reshaping the field of pharmaceutical waste management. By integrating thorough depackaging solutions, healthcare facilities can meet regulatory standards, cut down on expenses, and lessen their environmental impact. Investing in proper pharmaceutical waste management is more than just a regulatory requirement—it’s a pledge to protect the environment and public health.

